Radioactive emission
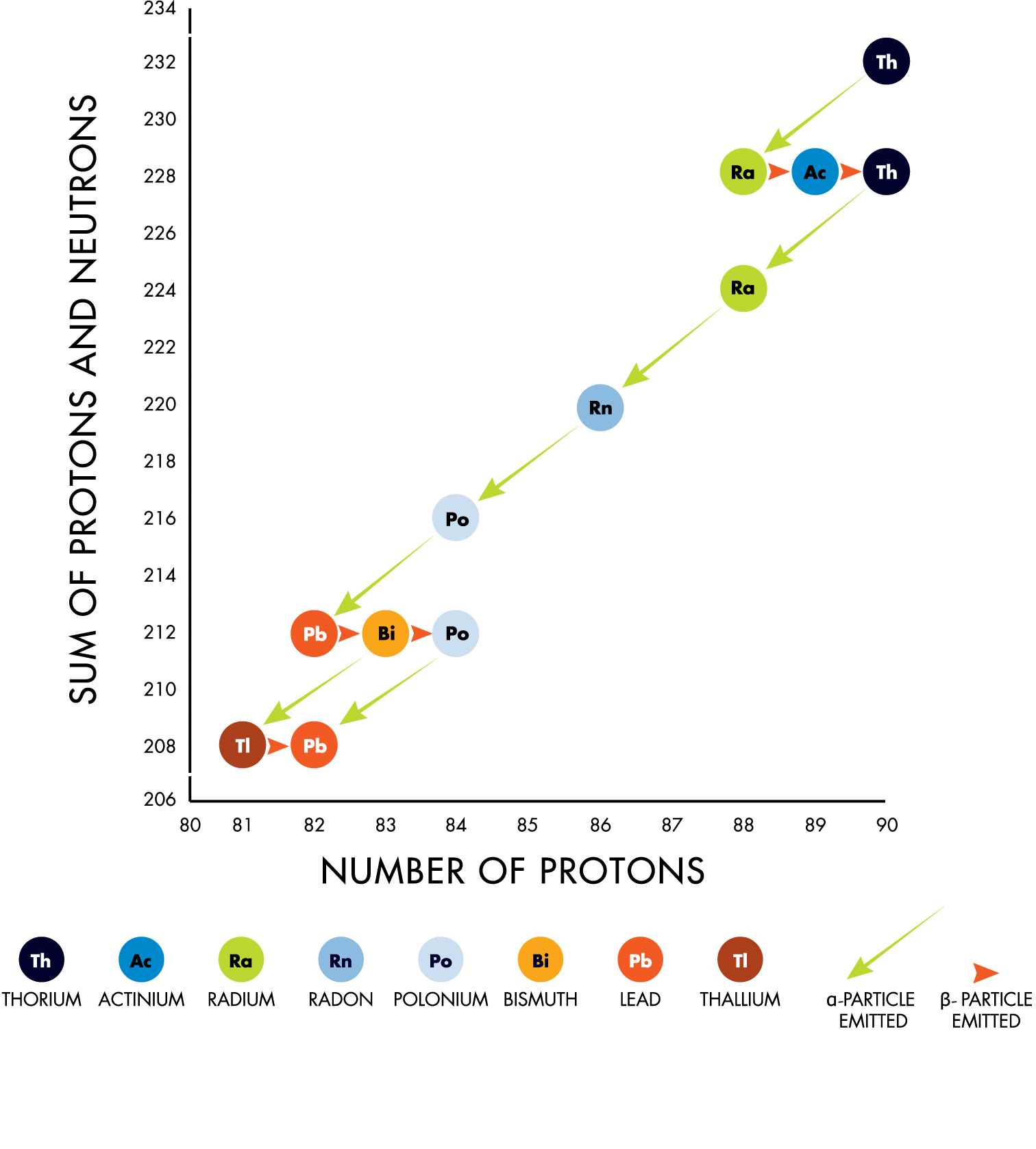
An atom of a radioactive material can emit alpha particles (helium nuclei) or can undergo beta decay, where a neutron is replaced by a proton, with accompanying emission of a beta particle (electron) and an electron-type antineutrino. Both processes change the atom’s chemical identity. The new atom may also be radioactive, leading to decay chains that end when a stable isotope is reached, for example lead-208.
Historically, studies using alpha particles were crucial to establishing the nuclear model of the atom. In the early 1930s, analysis of the energy spectrum of beta particles led Wolfgang Pauli and Enrico Fermi to a theory requiring the existence of neutrinos. These were first detected experimentally in 1956.
At the Large Hadron Collider, particles from radioactive sources are used to calibrate the response of detector components.







