Into the atom
The idea that matter is made of particles was given strong scientific foundations in the nineteenth century, through the work of John Dalton, Amedeo Avogadro, Robert Brown and others. The basic unit of matter from a chemical perspective was given the name atom, from the Greek ἄτομος (átomos), meaning indivisible. The name was inaccurate, and the first atomic constituent, the electron, was discovered by J.J. Thomson in 1896. A year earlier, Henri Becquerel had found that certain materials spontaneously emitted radiation. Work by Ernest Rutherford and Paul Villard divided this radiation into three types: alpha particles (now known to consist of two protons and two neutrons), beta particles (electrons) and gamma rays (photons).
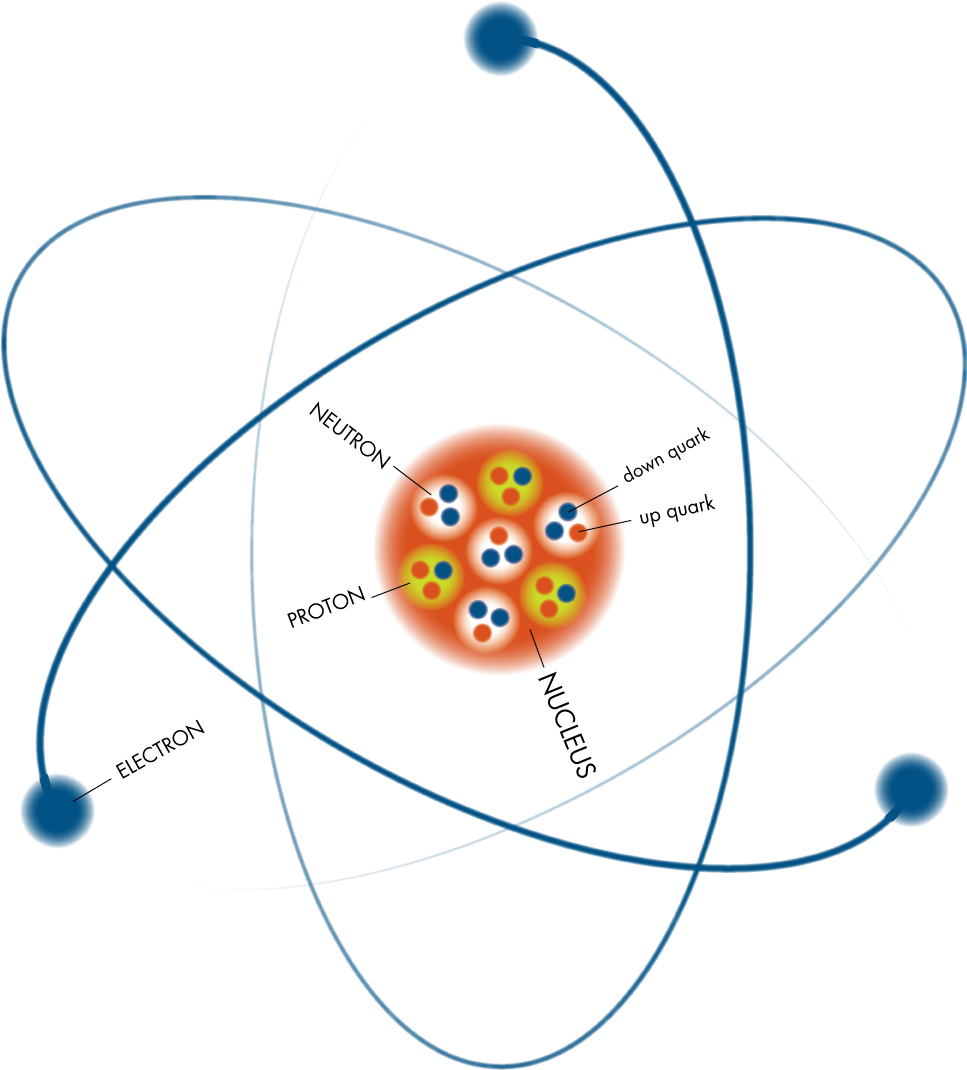
In an experiment performed in 1909, Hans Geiger and Ernest Marsden found that alpha particles fi red at gold foil could be scattered through large angles. Rutherford, who supervised the work, thought the result astonishing: “almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.” An analysis of the measurements led, in 1911, to his model of the atom as a small, dense, positively charged nucleus, orbitted by negatively charged electrons. Subsequent studies showed that the atomic nucleus is built from protons and neutrons, collectively known as nucleons. Rutherford's demonstration, in 1919, that the hydrogen nucleus is contained in other nuclei can be regarded as the discovery of the proton. The neutron was discovered by James Chadwick, in 1932.
The number of protons in the nucleus of an atom is exactly equal to the number of orbitting electrons, and defi nes the atom's chemical identity. Materials consisting of atoms that all have the same number of protons are termed elements. Atoms of an element that contain different numbers of neutrons are said to be isotopes of the element, and are labelled by the total number of protons and neutrons. For example, carbon-12 contains 6 protons and 6 neutrons; carbon-14 contains 6 protons and 8 neutrons. An atom that gains or loses electrons, so becoming electrically charged, is known as an ion. The physical process that causes ions to form is ionisation.
Experiments in the late 1960s and early 1970s to study the scattering of high-energy leptons by nucleons provided fi rst evidence that nucleons are themselves composite particles. The model today is that a proton is made from two up quarks and one down quark, and a neutron is made from two down quarks and one up quark. The quarks in each nucleon are strongly bound by gluons.







